Unit Three: Chemical Bonding
This unit will allow you to explore the four types of chemical bonds. These are covalent, ionic, metallic, and hydrogen bonding. Furthermore, this unit will explain how to draw out the Lewis Structures for each molecule as well as the three dimensional shapes associated with the molecules. This section will end with basic nomenclature of ionic and molecular compounds.
Lesson One: Basic Concepts
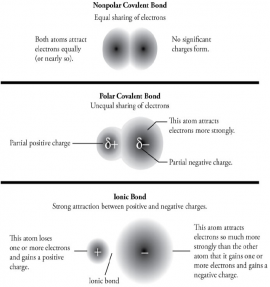
In this lesson, you will be able to understand the beginning concepts of chemical bonding. Based upon valence electrons determined through the electron configuration of atoms, you should understand how atoms are able to bond together. Furthermore, you will be able to distinguish between covalent and ionic chemical bonds based upon electronegativity.
Lesson Two: Covalent Bonding
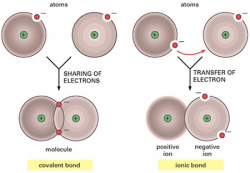
This lesson is based upon the basic principles of covalent bonding. After this lesson you should be able to understand the sharing of electrons between atoms. Using the electronegativity tables, you should also recognize the difference between polar and nonpolar bonds.
Lesson Three: Lewis Structures
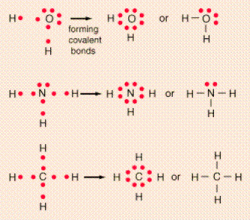
This lesson will introduce the idea of lewis structures and how they represent how the atoms are bonded together. After this lesson, you should be able to use the steps provided to create structures of basic molecules. Online tutorials will be used in this section to start to provide you with external sources of communication with the internet.
Lesson Four: Lewis Structures II
This lesson will continue to explain more complicated patterns of Lewis Structures. In this lesson, multiple bonds will be covered. These are double and triple bonds. Furthermore, this lesson will focus on more complicated Lewis Structures and provide extra practice through more tutorial websites that will be introduced. This lesson is self-directed so practice.
Lesson Five: Webquest
This lesson will help you to become more familiar with a bunch of new chemistry sites to add to your list of reputable sites. This lesson is designed to be self-taught self-motivated as you will be teaching the material to each other in a form of a PowerPoint presentation. Work with the wikispace link attached to complete the assignment. This lesson is designed to be a research period.
Lesson Six: Introduction to VSEPR
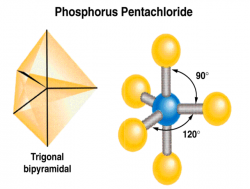
This lesson is designed to introduce another use of the Lewis Structures to form the three dimensional molecular geometries of molecules. This lesson will practice making Lewis structures into a more accurate image of the molecule based on the rules outlined in the VSEPR theory.
Lesson Seven: Bond Lengths and Angles in VSEPR Shapes
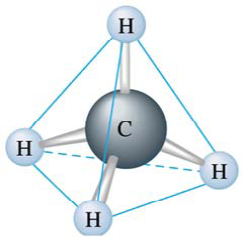
This lesson will work off of the previous lesson and continue to go into detail about the many three dimensional molecular shapes. The goal of this lesson is to teach the students about how VSEPR structures and the theories also work to determine and predict bond angles and shapes within molecules.
Lesson Eight: Heat of Reaction Experiment
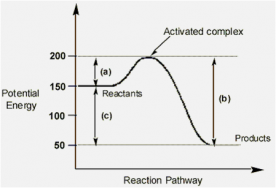
In this lesson you will complete the lab outline in the lesson page. This lab will require you to work in partners and work with heats of reaction between acids and bases and common household ingredients.
Lesson Nine: Nomenclature

This lesson is designed for you to become familiar with names of inorganic and organic compounds. You will learn to make the chemical formulas and use nomenclature to indicate the molecular or ionic compounds.
